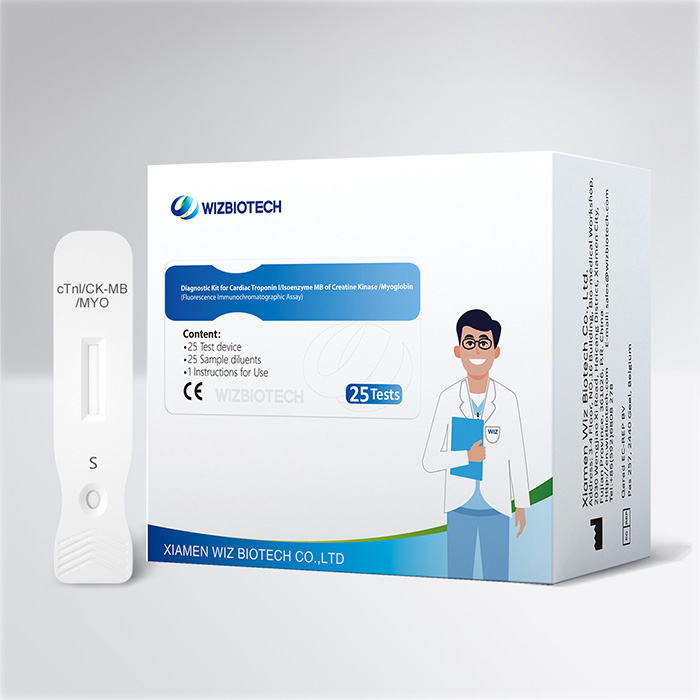
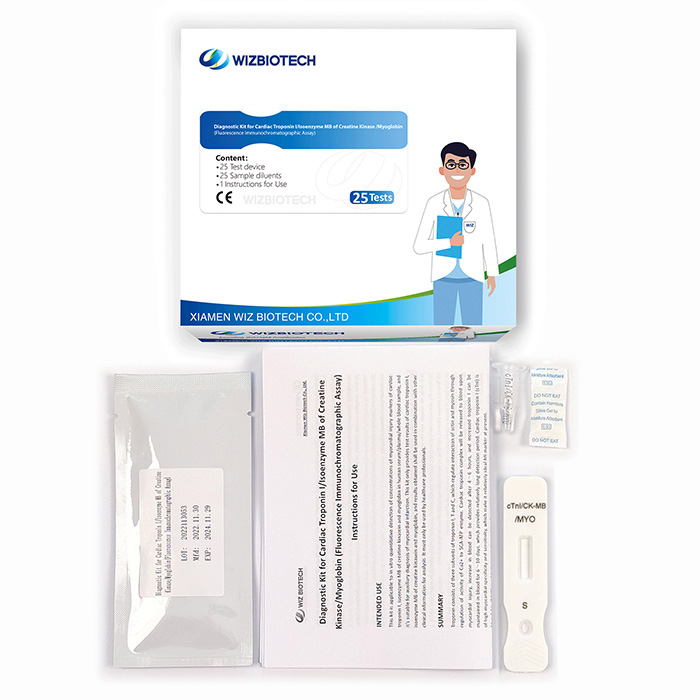
CK-MB/cTnI/Myo Fast Test Kit (Immunofluorescence Assay) For Myocardial Infarction (MI) And Myocardial Injury

- Wizbiotech
- CE, UKCA
- China
This kit is applicable to in vitro quantitative detection of concentrations of myocardial injury markers of cardiac troponin I, isoenzyme MB of creatine kinasein and myoglobin in human serum/plasma/whole blood sample, and it's suitable for auxiliary diagnosis of myocardial infarction.
Benefits
Very simple to use.In clinical trials, it is more beneficial to speed up the process of diagnosing a patient's disease.
Quick result.The test results were obtained in 15 minutes, which met the consensus requirements of the guidelines.
quantification.
Clinical Significance
Auxiliary diagnosis of myocardial infarction
Evaluate the effect of thrombolytic therapy
Product Performance
Reference method vs cTnl/CK-MB/MYO | |
Correlation with IFA | Y=0.975X+0.074,R=0.9854(cTnl) Y=0.915X+0.242,R=0.9885(CK-MB) Y=0.989X+2.759,R=0.9897(MYO) |
CV | ≤15% |
Differ | Within 15% |
Application
Outpatient Emergency Laboratory
Clinical Departments
Community Hospital
Laboratory Departments
Health Management Center
Clinic
Certifications